Uridine diphosphate N-acetylglucosamine
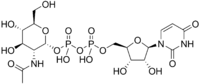 | |
| Names | |
|---|---|
| IUPAC name
[(2R,3R,4R,5S,6R)-3-acetamido-4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl] [[(2R,3S,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl] hydrogen phosphate | |
| Other names
UDP-N-acetylglucosamine; UDP-GlcNAc | |
| Identifiers | |
| 91183-98-1 | |
| 3D model (Jmol) | Interactive image |
| 1779 | |
| PubChem | 445675 |
| |
| Properties | |
| C17H27N3O17P2 | |
| Molar mass | 607.35 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Uridine diphosphate N-acetylglucosamine or UDP-GlcNAc is a nucleotide sugar and a coenzyme in metabolism. It is used by glycosyltransferases to transfer N-acetylglucosamine residues to substrates. D-Glucosamine is made naturally in the form of glucosamine-6-phosphate, and is the biochemical precursor of all nitrogen-containing sugars.[1] To be specific, glucosamine-6-phosphate is synthesized from fructose 6-phosphate and glutamine[2] as the first step of the hexosamine biosynthesis pathway.[3] The end-product of this pathway is UDP-GlcNAc, which is then used for making glycosaminoglycans, proteoglycans, and glycolipids.[4]
UDP-GlcNAc is extensively involved in intracellular signaling as a substrate for O-linked N-acetylglucosamine transferases (OGTs) in a wide range of species. It is also involved in nuclear pore formation and nuclear signalling. OGTs and OG-ases play an important role in the structure of the cytoskeleton. In mammals, there is enrichment of OGT transcripts in the pancreas beta-cells, and UDP-GlcNAc is thought to be part of the glucose sensing mechanism. There is also evidence that it plays a part in insulin sensitivity in other cells. In plants, it is involved in the control of gibberellin production.[5]
Clostridium novyi type A alpha-toxin is an O-linked N-actetylglucosamine transferase acting on Rho proteins and causing the collapse of the cytoskeleton.
References
- ↑ Roseman, S (2001). "Reflections on glycobiology". The Journal of Biological Chemistry. 276 (45): 41527–42. doi:10.1074/jbc.R100053200. PMID 11553646.
- ↑ Sudhamoy Ghosh; Blumenthal, HJ; Davidson, E; Roseman, S (1960-05-01). "Glucosamine Metabolism". Journal of Biological Chemistry. 235 (5): 1265–73. PMID 13827775.
- ↑ International Union of Biochemistry and Molecular Biology
- ↑ Milewski S, Gabriel I, Olchowy J (2006). "Enzymes of UDP-GlcNAc biosynthesis in yeast". Yeast. 23 (1): 1–14. doi:10.1002/yea.1337. PMID 16408321.
- ↑ Hanover, J. A. (2001). "Glycan-dependent signaling: O-linked N-acetylglucosamine". The FASEB Journal. 15: 1865–1876. doi:10.1096/fj.01-0094rev.