Mebhydrolin
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral[1] |
| ATC code | R06AX15 (WHO) |
| Identifiers | |
| |
| Synonyms | 9-Benzyl-2-methyl-2,3,4,9-tetrahydro-1H-gamma-carboline, Incidal, Omeril, Diazolin, Fabahistin, mebhydrolin napadisylate, mebhydroline 1,5-naphthalenedisulfonate[2] |
| CAS Number |
524-81-2 |
| PubChem (CID) | 22530 |
| ChemSpider |
21129 |
| UNII |
9SUK9B7XVY |
| KEGG |
D08161 |
| ECHA InfoCard | 100.007.606 |
| Chemical and physical data | |
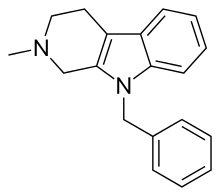
| Formula | C19H20N2[4] |
| Molar mass | 276.376 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Mebhydrolin (INN) or mebhydroline is an antihistamine. It is not available in the United States, but it is in various other countries under the brand names Bexidal (BD) and Diazolin (RU). It is used for symptomatic relief of allergic symptoms caused by histamine release, including nasal allergies and allergic dermatosis.
Mebhydrolin has been shown to magnify the performance-deficit effects of alcohol.[5]
References
- 1 2 "FABAHISTIN 50 mg (Tablets)". South African Electronic Package Inserts. September 1970. Archived from the original on October 17, 2006. Retrieved 2007-03-02.
- ↑ "Mebhydroline". National Library of Medicine - Medical Subject Headings. US National Institutes of Health, National Library of Medicine. Retrieved 2007-03-02.
- ↑ "Diazoline". National Library of Medicine - Medical Subject Headings. US National Institutes of Health, National Library of Medicine. Retrieved 2007-03-02.
- ↑ "Mebhydrolin chemical information". PubChem. Retrieved 2007-03-02.
- ↑ Franks HM, Lawrie M, Schabinsky VV, Starmer GA, Teo RK (October 1981). "Interaction between ethanol and antihistamines: 3. mebhydrolin". Med. J. Aust. 2 (9): 477–9. PMID 6119605.
This article is issued from Wikipedia - version of the 9/26/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.