17β-Hydroxysteroid dehydrogenase
| 17β-Hydroxysteroid dehydrogenase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 1.1.1.51 | ||||||||
| CAS number | 9015-81-0 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / EGO | ||||||||
| |||||||||
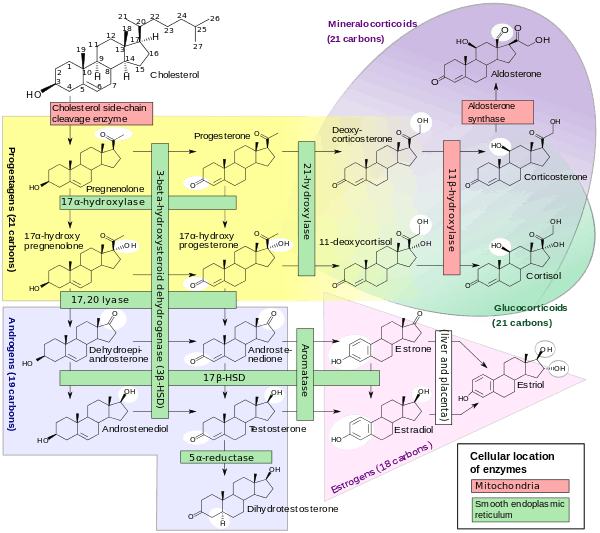
17β-Hydroxysteroid dehydrogenases (EC 1.1.1.51, beta-hydroxy steroid dehydrogenase, 17-ketoreductase, 17beta-hydroxy steroid dehydrogenase, 3beta-hydroxysteroid dehydrogenase, 3beta-hydroxy steroid dehydrogenase, 17β-HSD, 17-ketosteroid oxidoreductases, HSD17B, 17-ketosteroid reductases, 17-KSR), are a group of alcohol oxidoreductases which catalyse the dehydrogenation of 17-hydroxysteroids in steroidogenesis.[1][2][3][4][5] This includes interconversion of DHEA and androstenediol, androstenedione and testosterone, and estrone and estradiol, respectively.[6][7]
Note that the major reactions catalysed by 17β-HSD (e.g., the conversion of androstenedione to testosterone) are in fact hydrogenation (reduction) rather than dehydrogenation (oxidation) reactions.
Genes
Genes coding for 17β-HSD include:
- HSD17B1
- HSD17B2
- HSD17B3
- HSD17B4
- HSD17B6
- HSD17B7
- HSD17B8
- HSD17B10
- HSD17B11
- HSD17B12
- HSD17B13
- HSD17B14
Clinical significance
Isozyme 3 is responsible for 17-beta-hydroxysteroid dehydrogenase deficiency.
Some inhibitors of 17(-hydroxysteroid dehydrogenase type 1 were identified, for example esters of cinnamic acid and various flavones (e.g. Fisetin).[8]
References
- ↑ Dahm, K.; Breuer, H. (1964). "Anreicherung einer 17β-hydroxysteroid:NAD(P)-oxydoreduktase aus der Nebenniere der Ratte". Hoppe-Seyler's Z. Physiol. Chem. 336: 63–68. doi:10.1515/bchm2.1964.336.1.63. PMID 14214322.
- ↑ Lynn, W. S.; Brown, R. H. (1958). "The conversion of progesterone to androgens by testes". J. Biol. Chem. 232 (2): 1015–1030. PMID 13549484.
- ↑ Marcus, P. I.; Talalay, P. (1956). "Induction and purification of α- and β-hydroxysteroid dehydrogenases". J. Biol. Chem. 218 (2): 661–674. PMID 13295221.
- ↑ Schultz, R. M.; Groman, F. V.; Engel, L. L. (1977). "3(17)β-Hydroxysteroid dehydrogenase of Pseudomonas testosteroni. A convenient purification and demonstration of multiple molecular forms". J. Biol. Chem. 252: 3775–3783. PMID 193845.
- ↑ Talalay, P.; Dobson, M. M. (1953). "Purification and properties of a α-hydroxysteroid dehydrogenase". J. Biol. Chem. 205: 823–837. PMID 13129261.
- ↑ Labrie, F.; Luu-The, V.; Lin. S. X.; et al. (January 1997). "The key role of 17 beta-hydroxysteroid dehydrogenases in sex steroid biology". Steroids. 62 (1): 148–58. doi:10.1016/S0039-128X(96)00174-2. PMID 9029730.
- ↑ Charles G. D. Brook; Daniel Truong, M.D.; Peter Clayton; William Carroll; Rosalind Brown (22 September 2011). Brook's Clinical Pediatric Endocrinology. John Wiley & Sons. p. 288. ISBN 978-1-4443-1673-5. Retrieved 29 April 2012.
- ↑ Brožič, Petra; Kocbek, Petra; Sova, Matej; Kristl, Julijana; Martens, Stefan; Adamski, Jerzy; Gobec, Stanislav; Lanišnik Rižner, Tea (2009). "Flavonoids and cinnamic acid derivatives as inhibitors of 17β-hydroxysteroid dehydrogenase type 1". Molecular and Cellular Endocrinology. 301 (1-2): 229–234. doi:10.1016/j.mce.2008.09.004. ISSN 0303-7207.
- 17 beta-Hydroxysteroid Dehydrogenases at the US National Library of Medicine Medical Subject Headings (MeSH)
External links
- 3(or 17)beta-hydroxysteroid dehydrogenase at the US National Library of Medicine Medical Subject Headings (MeSH)