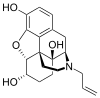
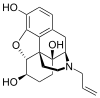
Naloxol
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
α-naloxol: (4R,4aS,7S,7aR,12bS)-3-allyl-2,3,4,4a,5,6,7,7a-octahydro-1H-4,12-methanobenzofuro[3,2-e]isoquinoline-4a,7,9-triol β-naloxol: (4R,4aS,7R,7aR,12bS)-3-allyl-2,3,4,4a,5,6,7,7a-octahydro-1H-4,12-methanobenzofuro[3,2-e]isoquinoline-4a,7,9-triol | |||
| Identifiers | |||
| 20410-95-1 (α) 53154-12-4 (β) 58691-01-3 (α/β) | |||
| 3D model (Jmol) | (α): Interactive image (β): Interactive image | ||
| ChemSpider | 4590928 (α) 9177198 (β) | ||
| ECHA InfoCard | 100.236.696 | ||
| PubChem | 5492271 (α) 5492293 (β) | ||
| |||
| |||
| Properties | |||
| C19H23NO4 | |||
| Molar mass | 329.40 g·mol−1 | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Naloxol is an opioid antagonist closely related to naloxone. It exists in two isomeric forms, α-naloxol and β-naloxol.
α-naloxol is a human metabolite of naloxone.[1] Synthetically, α-naloxol can be prepared from naloxone by reduction of the ketone group, and β-naloxol can be prepared from α-naloxol by a Mitsunobu reaction.[2]
Naloxol can be said to be the oxymorphol analogue of naloxone.
See also
References
- ↑ Weinstein, S. H.; Pfeffer, M.; Schor, J. M.; Indindoli, L.; Mintz, M. (1971). "Metabolites of naloxone in human urine". Journal of Pharmaceutical Sciences. 60 (10): 1567–1568. doi:10.1002/jps.2600601030. PMID 5129377.
- ↑ Simon, C (1994). "Stereoselective synthesis of β-naltrexol, β-naloxol β-naloxamine, β-naltrexamine and related compounds by the application of the mitsunobu reac". Tetrahedron. 50 (32): 9757. doi:10.1016/S0040-4020(01)85541-1.
This article is issued from Wikipedia - version of the 6/4/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.