Pasireotide
 | |
| Clinical data | |
|---|---|
| Trade names | Signifor |
| License data | |
| Routes of administration | Subcutaneous |
| ATC code | H01CB05 (WHO) |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| Synonyms | SOM230 |
| CAS Number |
396091-73-9 |
| PubChem (CID) | 9941444 |
| IUPHAR/BPS | 2018 |
| DrugBank |
DB06663 |
| ChemSpider |
8117062 |
| UNII |
98H1T17066 |
| KEGG |
D10147 |
| ChEBI |
CHEBI:72312 |
| ECHA InfoCard | 100.211.883 |
| Chemical and physical data | |
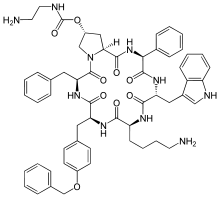
| Formula | C58H66N10O9 |
| Molar mass | 1047.21 g/mol[1] |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Pasireotide (SOM230, trade name Signifor[2]) is an orphan drug approved in the United States[3] and Europe[4] for the treatment of Cushing's disease in patients who fail or are ineligible for surgical therapy.[5][6] It was developed by Novartis. Pasireotide is a somatostatin analog with a 40-fold increased affinity to somatostatin receptor 5 compared to other somatostatin analogs.
Pasireotide was approved for Cushing's disease by the EMEA in October 2009[7] and by the FDA in December 2012.[8]
Pasireotide LAR was approved by the FDA for treatment of acromegaly in December 2014, and had been approved for this indication by the EMEA one month earlier.[9]
References
- ↑ Pasireotide | C58H66N10O9 - PubChem http://pubchem.ncbi.nlm.nih.gov/compound/Pasireotide
- ↑ Signifor® (pasireotide) Official Website for healthcare professionals outside the US http://www.signifor.com/
- ↑ http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/200677lbl.pdf
- ↑ http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/002052/WC500128056.pdf
- ↑ "Novartis drug Signifor® approved in the EU as the first medication to treat patients with Cushing's disease". Retrieved 2012-07-08.
- ↑ Mancini, Tatiana; Porcelli, Teresa; Giustina, Andrea (2010). "Treatment of Cushing disease: overview and recent findings". Therapeutics and Clinical Risk Management. 6: 505–16. doi:10.2147/TCRM.S12952 (inactive 2015-02-01). PMC 2963160
 . PMID 21063461.
. PMID 21063461. - ↑ EMEA Approval for Pasireotide
- ↑ "FDA Approves Pasireotide for Cushing's Disease".
- ↑ Tucker, Miriam E. FDA Approves Pasireotide for Treating Acromegaly, Medscape, December 17, 2014, Retrieved 2015-08-21
This article is issued from Wikipedia - version of the 9/27/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.