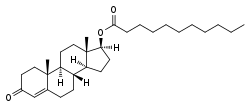
Testosterone undecanoate
 | |
| Clinical data | |
|---|---|
| Pronunciation | tess-toss-ter-own un-deck-ah-no-ate |
| Pregnancy category |
|
| Routes of administration | Oral, intramuscular injection |
| ATC code | G03BA03 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability |
Oral: 3–7% Intramuscular ~100% |
| Metabolism | Liver, testis and prostate |
| Biological half-life | 1–12 days |
| Excretion | Urine |
| Identifiers | |
| |
| Synonyms |
Aveed, Andriol, Undestor, Nebido, Pantestone, Restandol, Cernos Depot, Nebido-R, Reandron 1000 17β-[(1-Oxoundecyl)oxy]-androst-4-en-3-one |
| CAS Number |
5949-44-0 |
| PubChem (CID) | 65157 |
| ChemSpider |
58664 |
| UNII |
H16A5VCT9C |
| KEGG |
D06087 |
| ChEMBL |
CHEMBL2107067 |
| Chemical and physical data | |
| Formula | C30H48O3 |
| Molar mass | 456.70032 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Testosterone undecanoate (USAN, BAN) (brand names Aveed, Andriol, Androxon, Cernos Depot, Nebido, Panteston, Restandol, Nebido-R, Reandron 1000, Undestor), or testosterone undecylate, is an androgen and anabolic steroid and a testosterone ester.[1][2][3] It is used in androgen replacement therapy primarily for the treatment of male hypogonadism, and has also been investigated for use as a male contraceptive. Unlike other testosterone esters, testosterone undecanoate is available in both oral and intramuscular formulations.[4] The Reandron 1000 formulation (Nebido in the United States) contains 1000 mg of testosterone undecanoate suspended in castor oil with benzyl benzoate for solubilisation and as a preservative, and is administered by intramuscular injection. As an excipient, benzyl benzoate has been reported as a cause of anaphylaxis in a case in Australia.[5] Bayer includes this report in information for health professionals and recommends that physicians "should be aware of the potential for serious allergic reactions" to preparations of this type.[6] In Australia, reports to ADRAC, which evaluates reports of adverse drug reactions for the Therapeutic Goods Administration, show several reports of allergic issues since the anaphylaxis case from 2011.
Testosterone undecanoate has a very long elimination half-life and mean residence time when given as a depot intramuscular injection.[7] The elimination half-life and mean residence time of testosterone undecanoate are 2.5-fold and 4-fold longer than those of testosterone enanthate (the values for testosterone enanthate being 4.5 days and 8.5 days, respectively).[7]
Testosterone undecanoate is available in Europe but not in the United States.[8] It is administered at a dosage of 1,000 mg every 12 weeks via intramuscular injection.[9] In addition to parenteral, an oral formulation of testosterone undecanoate (brand name Andriol) is also available in Europe, but must be taken two or three times a day in combination with food.[9][10]
See also
References
- ↑ J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 641–642. ISBN 978-1-4757-2085-3.
- ↑ Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. ISBN 978-3-88763-075-1.
- ↑ I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. ISBN 978-94-011-4439-1.
- ↑ Köhn, Frank-Michael; Schill, Wolf-Bernhard (November 2003). "A new oral testosterone undecanoate formulation". World Journal of Urology. 21 (5): 311–315. doi:10.1007/s00345-003-0372-x. PMID 14579074.
- ↑ Ong, G. S. Y.; Somerville, C. P.; Jones, T. W.; Walsh, J. P. (2012). "Anaphylaxis Triggered by Benzyl Benzoate in a Preparation of Depot Testosterone Undecanoate". Case Rep Med. 2012. doi:10.1155/2012/384054. PMC 3261473
 . PMID 22272209. 384054.
. PMID 22272209. 384054. - ↑ "Nebido Monograph – Information for Health Care Professionals". Bayer. 2016. Retrieved 19 October 2016.
- 1 2 Anita H. Payne; Matthew P. Hardy (28 October 2007). The Leydig Cell in Health and Disease. Springer Science & Business Media. pp. 423–. ISBN 978-1-59745-453-7.
- ↑ Norman Lavin (28 March 2012). Manual of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 313–. ISBN 978-1-4511-4914-2.
- 1 2 S. Bertelloni; O. Hiort (28 September 2010). New Concepts for Human Disorders of Sexual Development. S. Karger AG. pp. 256–. ISBN 978-3-8055-9569-8.
- ↑ Jean L. Fourcroy (27 October 2008). Pharmacology, Doping and Sports: A Scientific Guide for Athletes, Coaches, Physicians, Scientists and Administrators. Routledge. pp. 25–. ISBN 978-1-134-08880-5.
External links
- Testosterone undecanoate at the US National Library of Medicine Medical Subject Headings (MeSH)