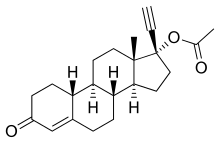
Norethisterone acetate
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Primolut-Nor, Aygestin, Gestakadin, Milligynon, Monogest, Norlutate, Primolut N, SH-420, Sovel, Styptin |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a604034 |
| Routes of administration | Oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| Synonyms | SH-420 |
| CAS Number |
38673-38-0 |
| PubChem (CID) | 63021 |
| ChemSpider |
5627 |
| ChEBI |
CHEBI:7628 |
| ChEMBL |
CHEMBL1201146 |
| Chemical and physical data | |
| Formula | C22H28O3 |
| Molar mass | 340.456 g/mol |
| | |
Norethisterone acetate (NETA) (INN, BAN), also known as norethindrone acetate (USAN), sold under brand names including Primolut-Nor (major), Aygestin (US), Gestakadin, Milligynon, Monogest, Norlutate (US, CA), Primolut N, SH-420 (UK), Sovel, Styptin, and others, is a steroidal progestin of the 19-nortestosterone group with additional weak androgenic and estrogenic activity[1][2] that is used orally as a hormonal contraceptive, in the treatment of gynecological disorders such as abnormal uterine bleeding, and as a component of hormone replacement therapy for menopause.[3][4][5] It is the 17β-acetate ester of norethisterone, and acts as a prodrug to norethisterone in the body.[6][7]
Pharmacology
Upon oral ingestion, NETA is rapidly converted into norethisterone by esterases during intestinal and first-pass hepatic metabolism.[8] Hence, it is a prodrug of norethisterone in the body.[6] As such, NETA is a potent progestogen with additional weak androgenic and estrogenic activity (the latter via its metabolite ethinyl estradiol).[1][2]
In terms of dosage equivalence, norethisterone and NETA are typically used at respective dosages of 0.35 mg/day and 0.6 mg/day as progestogen-only contraceptives, and at respective dosages of 0.5–1 mg/day and 1–1.5 mg/day in combination with ethinyl estradiol.[2] This suggests that NETA is around 50 to 66% as potent as norethisterone.[2] Conversely, the two drugs have been used at about the same dosages in hormone replacement therapy for menopausal symptoms.[2]
Chemistry
Synthesis
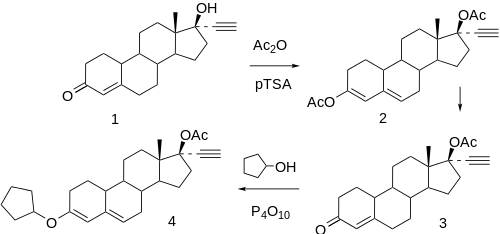
Reaction of norethindrone (1) with acetic anhydride in the presence of p-toluene sulfonic acid, affords norethindrone acetate (2).[9] This in turn affords, on reaction with exess cyclopentanol in the presence of excess phosphorus pentoxide, the 3-cyclopentyl enol ether (3),[10] the progestational component of Riglovic®.
History
Schering AG filed for a patent for NETA in June 1957, and the patent was issued in December 1960.[11] The drug was first marketed, by Parke-Davis as Norlestrin in the United States, in March 1964.[11][12] This was a combination formulation of 2.5 mg NETA and 50 μg ethinyl estradiol and was indicated as an oral contraceptive.[11][12] Other early brand names of NETA used in oral contraceptives included Minovlar and Anovlar.[11]
See also
References
- 1 2 Kuhl H (2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 Suppl 1: 3–63. doi:10.1080/13697130500148875. PMID 16112947.
- 1 2 3 4 5 IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; World Health Organization; International Agency for Research on Cancer (2007). Combined Estrogen-progestogen Contraceptives and Combined Estrogen-progestogen Menopausal Therapy. World Health Organization. pp. 417–. ISBN 978-92-832-1291-1.
Norethisterone and its acetate and enanthate esters are progestogens that have weak estrogenic and androgenic properties.
- ↑ F.. Macdonald (1997). Dictionary of Pharmacological Agents. CRC Press. p. 1450. ISBN 978-0-412-46630-4. Retrieved 12 May 2012.
- ↑ Index Nominum 2000: International Drug Directory. Taylor & Francis US. 2000. p. 750. ISBN 978-3-88763-075-1. Retrieved 30 May 2012.
- ↑ http://www.drugs.com/ppa/norethindrone-acetate.html
- 1 2 Thomas L. Lemke; David A. Williams (2008). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 1316–. ISBN 978-0-7817-6879-5.
- ↑ Norethisterone Acetate @ Drugs.com
- ↑ Chwalisz K, Surrey E, Stanczyk FZ (2012). "The hormonal profile of norethindrone acetate: rationale for add-back therapy with gonadotropin-releasing hormone agonists in women with endometriosis". Reprod Sci. 19 (6): 563–71. doi:10.1177/1933719112438061. PMID 22457429.
- 1 2 Iriarte, J.; Djerassi, C.; Ringold, H. J. (1959). "Steroids. CVII.1Δ5(6)-19-Nor Steroids, a New Class of Potent Anabolic Agents2". Journal of the American Chemical Society. 81 (2): 436. doi:10.1021/ja01511a042.
- 1 2 Ercoli, A.; Gardi, R. (1960). "Δ4-3-Keto Steroidal Enol Ethers. Paradoxical Dependency of Their Effectiveness on the Administration Route". Journal of the American Chemical Society. 82 (3): 746. doi:10.1021/ja01488a062.
- 1 2 3 4 Lara Marks (2010). Sexual Chemistry: A History of the Contraceptive Pill. Yale University Press. pp. 73–. ISBN 978-0-300-16791-7.
- 1 2 Robert W. Blum (22 October 2013). Adolescent Health Care: Clinical Issues. Elsevier Science. pp. 216–. ISBN 978-1-4832-7738-7.