Glucagon-like peptide-1
Glucagon-like peptide-1 (GLP-1) is a neuropeptide and an incretin derived from the transcription product of the proglucagon gene. The major source of GLP-1 in the periphery is the intestinal L cell that secretes GLP-1 as a gut hormone; the major source in the brain is the nucleus of the solitary tract, which is the source of a widely distributed set of GLP-1 projection neurons.[1] The biologically active forms of GLP-1 are: GLP-1-(7-37) and GLP-1-(7-36)NH2. Those peptides result from selective cleavage of the proglucagon molecule.
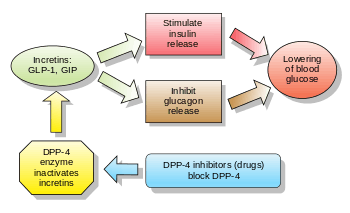
GLP-1 secretion by ileal L cells is dependent on the presence of nutrients in the lumen of the small intestine. The secretagogues (agents that cause or stimulate secretion) of this hormone include major nutrients like carbohydrate, protein and lipid. Once in the circulation, GLP-1 has a half-life of less than 2 minutes, due to rapid degradation by the enzyme dipeptidyl peptidase-4.
GLP-1 is a potent antihyperglycemic hormone, inducing the β-cells of the pancreas to release the hormone insulin in response to rising glucose, while suppressing glucagon secretion. Such glucose-dependent action is particularly attractive because an unregulated release of the hormone when the plasma glucose concentration is in the normal fasting range, or poorly-timed insulin injections, can cause a dangerous fall in blood glucose (hypoglycemia). This does not happen as a result of GLP-1 because GLP-1 no longer stimulates the β-cells to release more insulin when blood glucose levels are in the fasting range. Additionally, GLP-1 appears to restore the glucose sensitivity of pancreatic β-cells. The mechanism may involve increased expression of GLUT2 receptors on the surface of the β-cells; GLUT-2 is a carrier protein on the cell surface that facilitates the movement of plasma glucose across the cell membrane, allowing them to sense the level of glucose in circulation and release insulin when levels begin to climb. GLP-1 may also sensitize β-cells by increasing the expression of glucokinase, an enzyme in the β-cells that stimulates insulin production and release in response to glucose entering the cell. GLP-1 is also known to inhibit the programmed cell death (apoptosis) of pancreatic β-cells, and to stimulate β-cell proliferation and differentiation. In addition, GLP-1 inhibits gastric secretion and motility. This delays and protracts carbohydrate absorption and contributes to a satiating effect.
Physiological functions
Visceral functions
GLP-1 possesses several physiological properties that make it (and its functional analogs) a subject of intensive investigation as a potential treatment of diabetes mellitus.[2][3][4][5]
The known peripheral functions of GLP-1 include:
|
As evidence of the physiological role of GLP-1 in post-prandial insulin secretion, it has been shown that an oral dose of glucose triggers a much higher peak in plasma insulin concentration compared to an intravenous dose. Obese patients undergoing gastric bypass showed marked metabolic adaptations, resulting in frequent diabetes remission 1 year later. When the confounding of calorie restriction is factored out, β-cell function improves rapidly, very possibly under the influence of enhanced GLP-1 responsiveness.[7] Outside of its function as an insulin secretagogue, GLP-1 seems also to play a role in bone physiology. Researchers evidenced a massive reduction in bone strength in GLP-1 receptor knockout mice mainly due to a poor bone quality.[8]
CNS functions
The primary central nervous system functions of GLP-1 which are known include:[1][9]
|
|
Additional effects
There is evidence that GLP-1 has direct and indirect functions in the immune system.[10][11] Metabolic diseases (such as obesity and diabetes type II) are thought to have strong inflammatory underpinnings and vice versa, inflammation is often associated with metabolic changes.[12] GLP-1 might be one factor explaining this link, since its production seems to be altered under inflammatory conditions.[13]
See also
- Glucagon-like peptide 1 receptor
- Glucagon-like peptide-2
- Type 2 diabetes
- GLP-1 analogs : exenatide, liraglutide, dulaglutide
- Dipeptidyl peptidase-4
- Sitagliptin ( a DPP4 inhibitor)
- Liraglutide
- Glucose-dependent insulinotropic peptide
References
- 1 2 Skibicka KP (2013). "The central GLP-1: implications for food and drug reward". Front Neurosci. 7: 181. doi:10.3389/fnins.2013.00181. PMC 3796262
 . PMID 24133407.
. PMID 24133407. Much has been learned about the anatomical, neurochemical, and functional suppressive effects of GLP-1 or its analogs on food intake; GLP-1's ability to suppress food reward behavior is a new concept. ... The collective value of these findings is not only in offering support to the hypothesis that mesolimbic GLP-1 has a specific role in reward behavior but also in emphasizing the neuroanatomical separation of the reward and visceral illness mechanisms of GLP-1.
"Figure 1: Effect of GLP-1 on food intake and associated behaviors is neuroanatomicaly distributed" - ↑ "Diabetes and Intestinal Incretin Hormones: A New Therapeutic Paradigm" at medscape.com (slide 36)
- ↑ Toft-Nielsen MB, Madsbad S, Holst JJ (August 2001). "Determinants of the effectiveness of glucagon-like peptide-1 in type 2 diabetes". The Journal of Clinical Endocrinology and Metabolism. 86 (8): 3853–60. doi:10.1210/jcem.86.8.7743. PMID 11502823.
- ↑ Meier JJ, Weyhe D, Michaely M, et al. (March 2004). "Intravenous glucagon-like peptide 1 normalizes blood glucose after major surgery in patients with type 2 diabetes". Critical Care Medicine. 32 (3): 848–51. doi:10.1097/01.CCM.0000114811.60629.B5. PMID 15090972.
- ↑ de Graaf C, Donnelly C, Wootten D, et al. (2016). "Glucagon-Like Peptide-1 and Its Class B G Protein–Coupled Receptors: A Long March to Therapeutic Successes". Pharmacol Rev. 68 (4): 954–1013. doi:10.1124/pr.115.011395.
- ↑ Presswala L, Shubrook J (April 2015). "What to do after basal insulin: 3 Tx strategies for type 2 diabetes". The Journal of family practice. 64 (4): 214–20. PMID 25973447.
- ↑ Nannipieri M, Baldi S, Mari A, et al. (November 2013). "Roux-en-Y gastric bypass and sleeve gastrectomy: mechanisms of diabetes remission and role of gut hormones". The Journal of Clinical Endocrinology and Metabolism. 98 (11): 4391–9. doi:10.1210/jc.2013-2538. PMID 24057293.
- ↑ Mabilleau G, Mieczkowska A, Irwin N, Flatt PR, Chappard D (October 2013). "Optimal bone mechanical and material properties require a functional glucagon-like peptide-1 receptor". The Journal of Endocrinology. 219 (1): 59–68. doi:10.1530/JOE-13-0146. PMID 23911987.
- ↑
- de Graaf C, Donnelly C, Wootten D, et al. (2016). "Glucagon-Like Peptide-1 and Its Class B G Protein–Coupled Receptors: A Long March to Therapeutic Successes". Pharmacol Rev. 68 (4): 954–1013. doi:10.1124/pr.115.011395.
- ↑ Hadjiyanni, I.; Siminovitch, K. A.; Danska, J. S.; Drucker, D. J. "Glucagon-like peptide-1 receptor signalling selectively regulates murine lymphocyte proliferation and maintenance of peripheral regulatory T cells". Diabetologia. 53 (4): 730–740. doi:10.1007/s00125-009-1643-x.
- ↑ Rubin, Deborah C.; Zhang, Huiying; Qian, Peiqing; Lorenz, Robinna G.; Hutton, Karen; Peters, Marion G. (2000-10-15). "Altered enteroendocrine cell expression in T cell receptor alpha chain knock-out mice". Microscopy Research and Technique. 51 (2): 112–120. doi:10.1002/1097-0029(20001015)51:23.0.CO;2-D. ISSN 1097-0029.
- ↑ Rath, Eva; Haller, Dirk (2011-06-01). "Inflammation and cellular stress: a mechanistic link between immune-mediated and metabolically driven pathologies". European Journal of Nutrition. 50 (4): 219–233. doi:10.1007/s00394-011-0197-0. ISSN 1436-6215. PMID 21547407.
- ↑ Zietek, Tamara; Rath, Eva (2016-01-01). "Inflammation Meets Metabolic Disease: Gut Feeling Mediated by GLP-1". Inflammation: 154. doi:10.3389/fimmu.2016.00154. PMC 4840214
 . PMID 27148273.
. PMID 27148273.
External links
- Banting and Best Diabetes Centre at UT glp1
- Glucagon-Like Peptide 1 at the US National Library of Medicine Medical Subject Headings (MeSH)
- Insulin release pathways
American diabetes association:link-http://diabetes.diabetesjournals.org/content/56/1/8.full